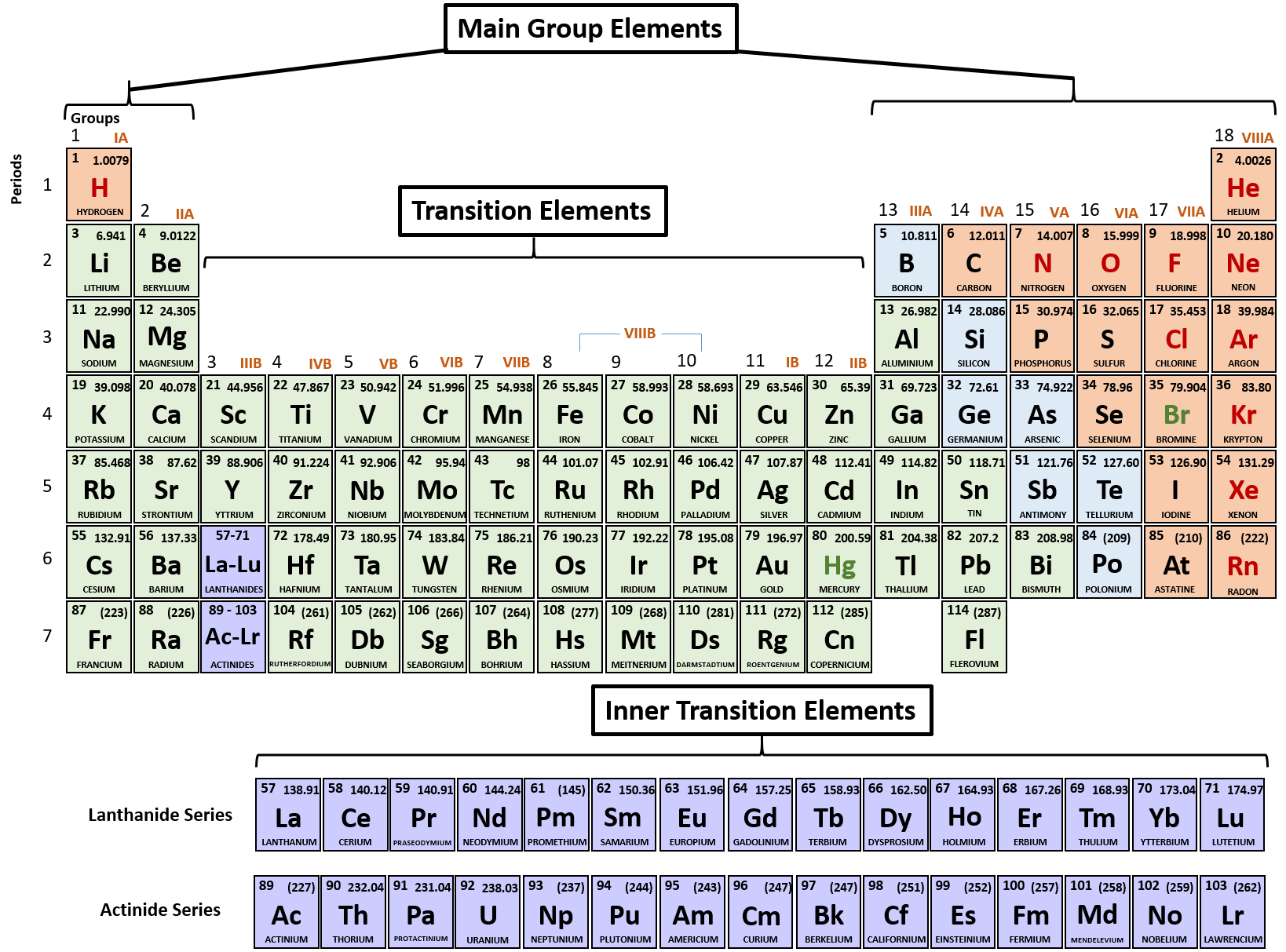
Some isotopes are less stable than others and, the less stable they are, the less likely that they are occur less in nature.Īs you can see, protium, the hydrogen with one proton only, is the most commonly occurring, and, as such, the mass number of hydrogen is usually written as 1. Isotopes are the variations of an elements, given by differing numbers of neutrons. You might have heard the term “ isotope” before. The Periodic Table of Elementsįirst, we should establish a familiarity with the periodic table. But, of course, I’ll show you what I mean. I don’t exaggerate when I say that, if you understand those two things, you understand most of orbital theory. In both lessons, we have discussed that modeling exactly where the electron is is difficult, but telling where it might be in a “cloud” of possible positions is far easier. The second furthered that discussion, showing that microscopic particles exhibit both particle-like behavior and wave-like behavior, changing the number of possible places that electrons can exist within the three dimensions, again, to minimize energy. The first lesson on electrons focused on their orientation and how they exist in a way that decreases their energy. Of course, if you want to understand these at a higher level, please check out part 1 and part 2. 
Orbitals are nothing more than the position of electrons in the space of an atom.īut, before we go over orbitals, let’s summarize the past lessons. For the rest of you, we have laid the groundwork for orbitals already by discussing what they are in detail. What if I told you that you already know what they are? For those of you who have already studied chemistry, you’ve already might have started to put together the pieces from the last two lessons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
